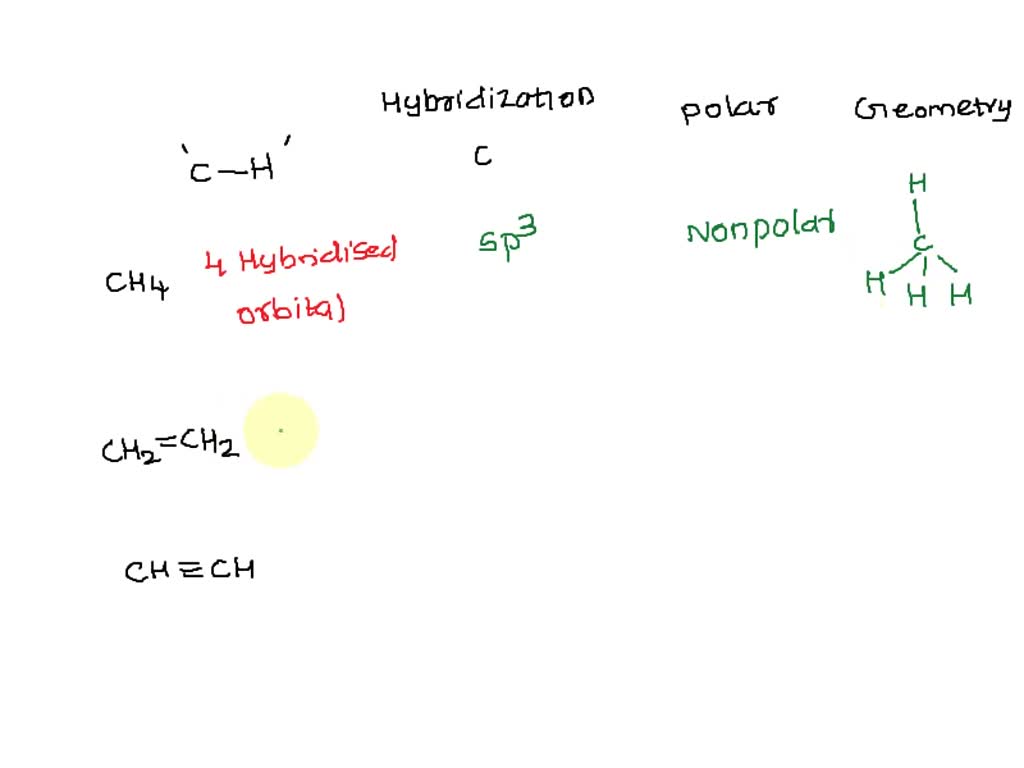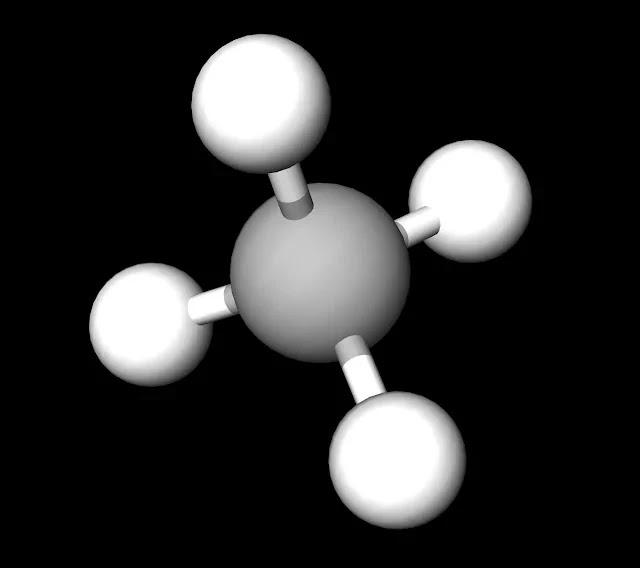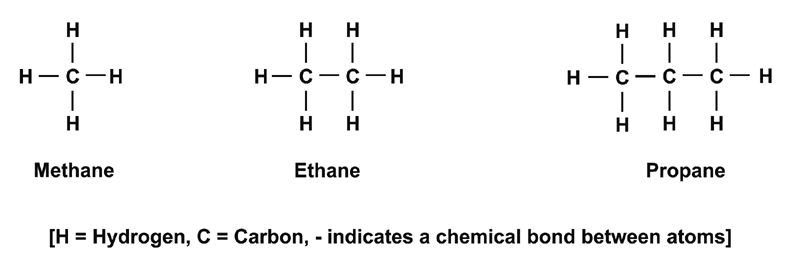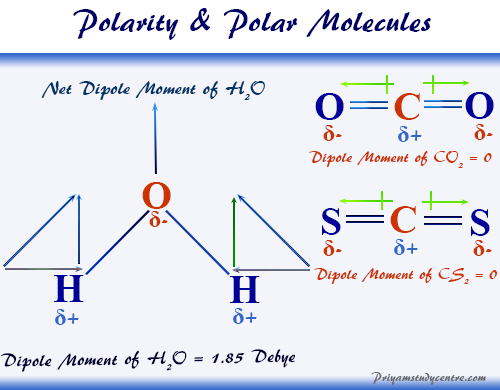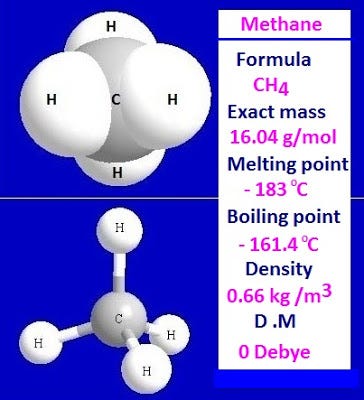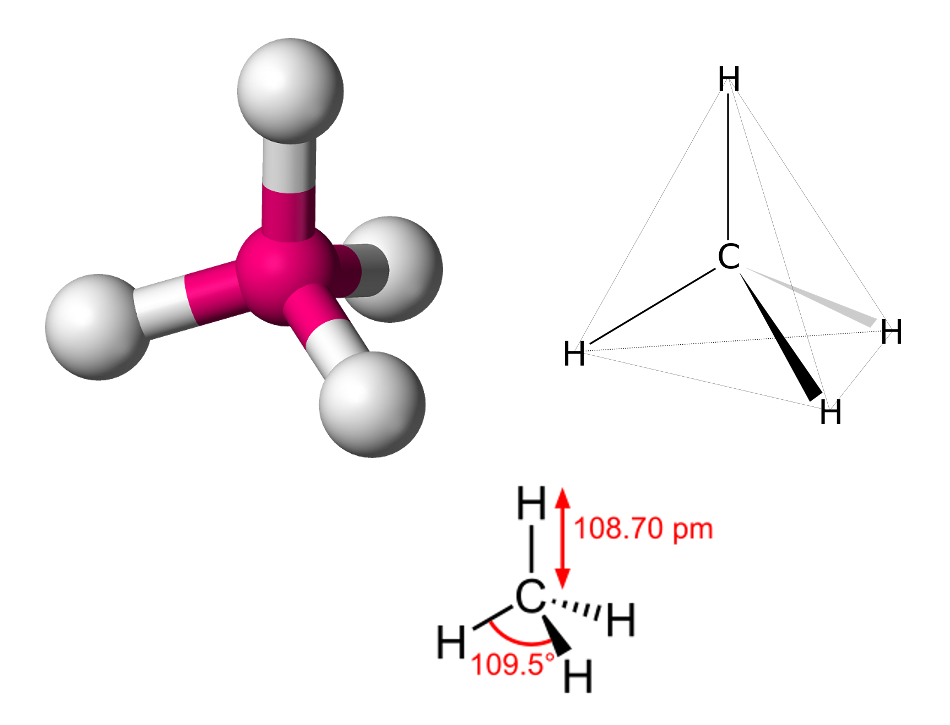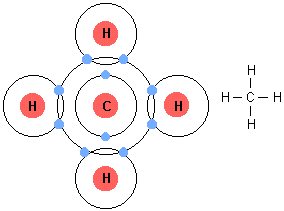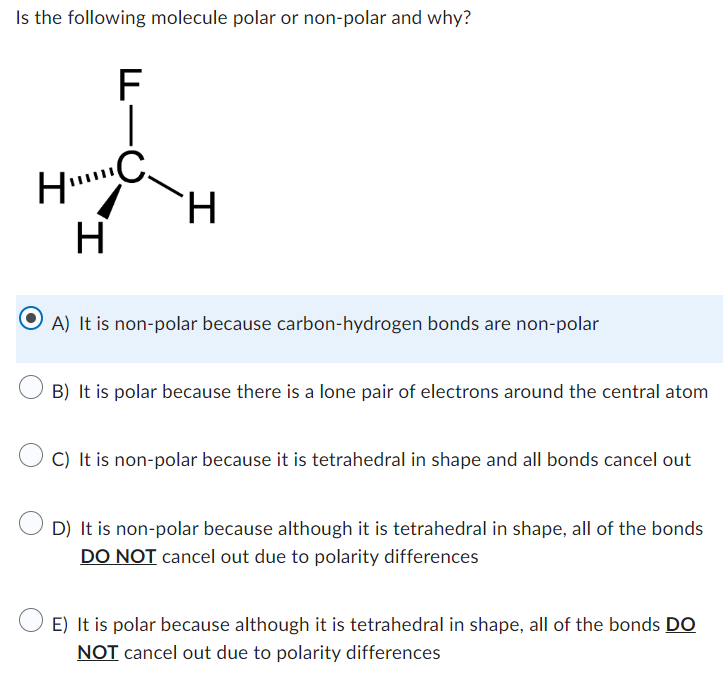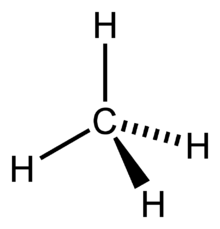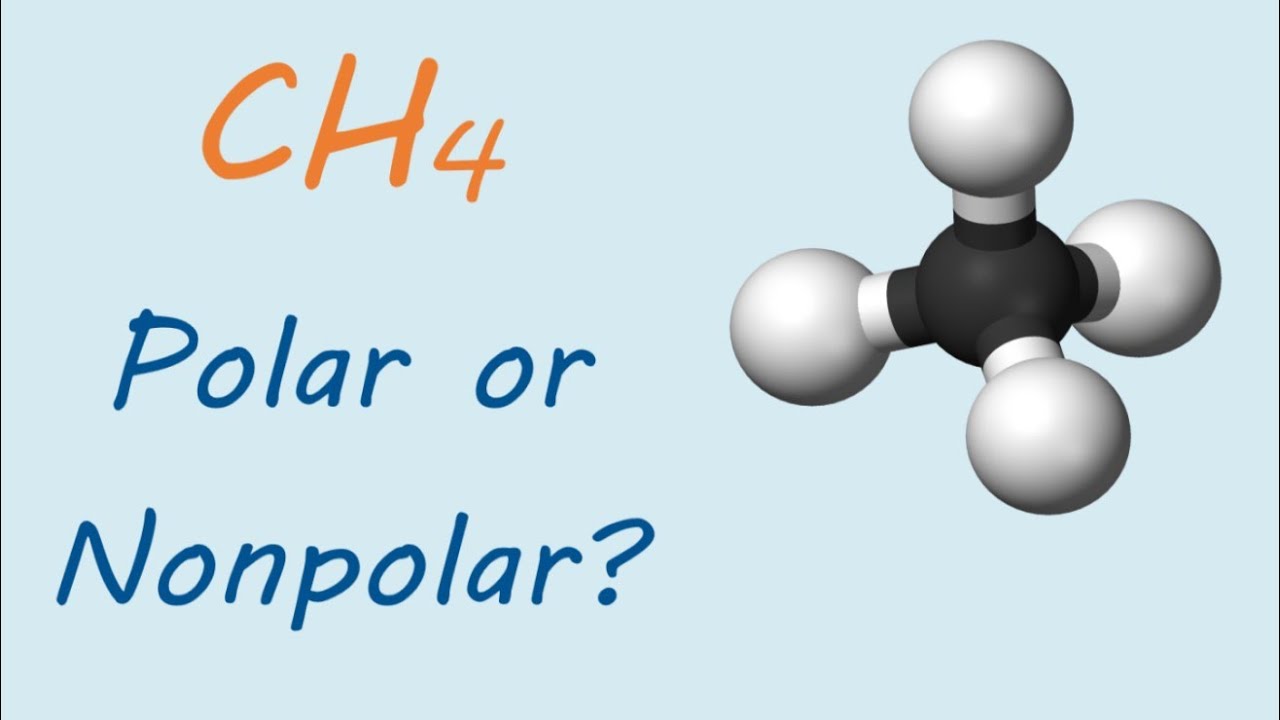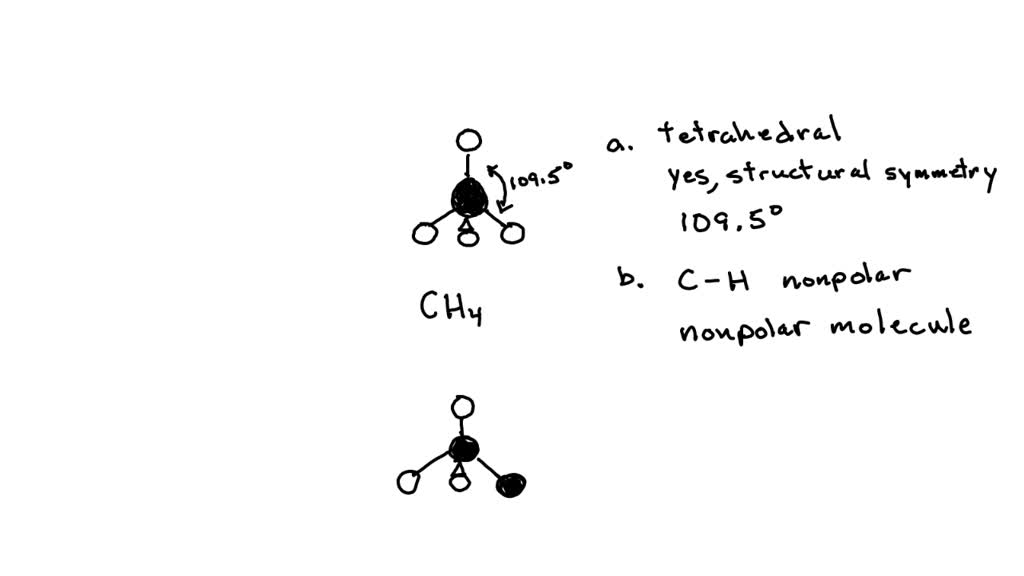
SOLVED: Build a molecule of methane, CH4 (a) Describe the molecule's overall shape, structural symmetry, and bond angles. (b) Is CH4 polar or nonpolar? 3. Replace one H atom with a C

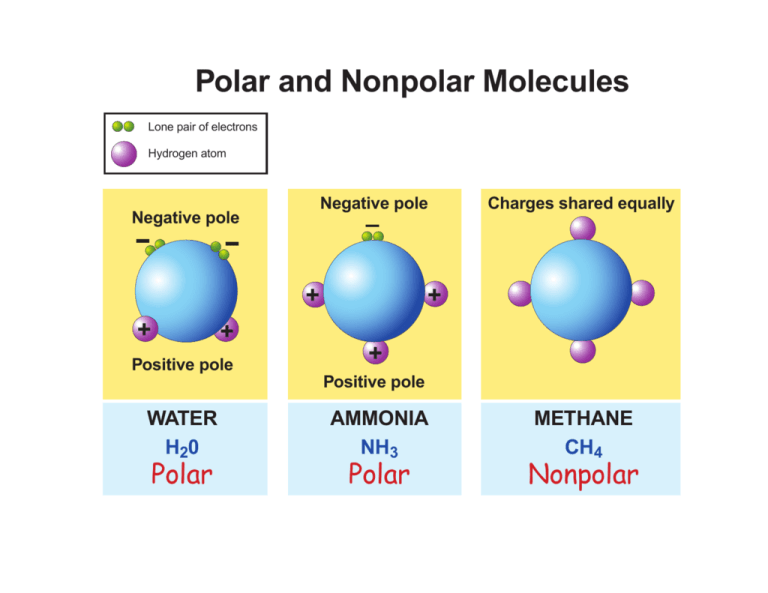
Research two common, polar molecules and two common nonpolar molecules. Draw their molecular structure and explain how the structure makes each molecule polar or non-polar | Homework.Study.com

PPT - Identify the following as ionic, polar covalent, or nonpolar covalent. PowerPoint Presentation - ID:2017614