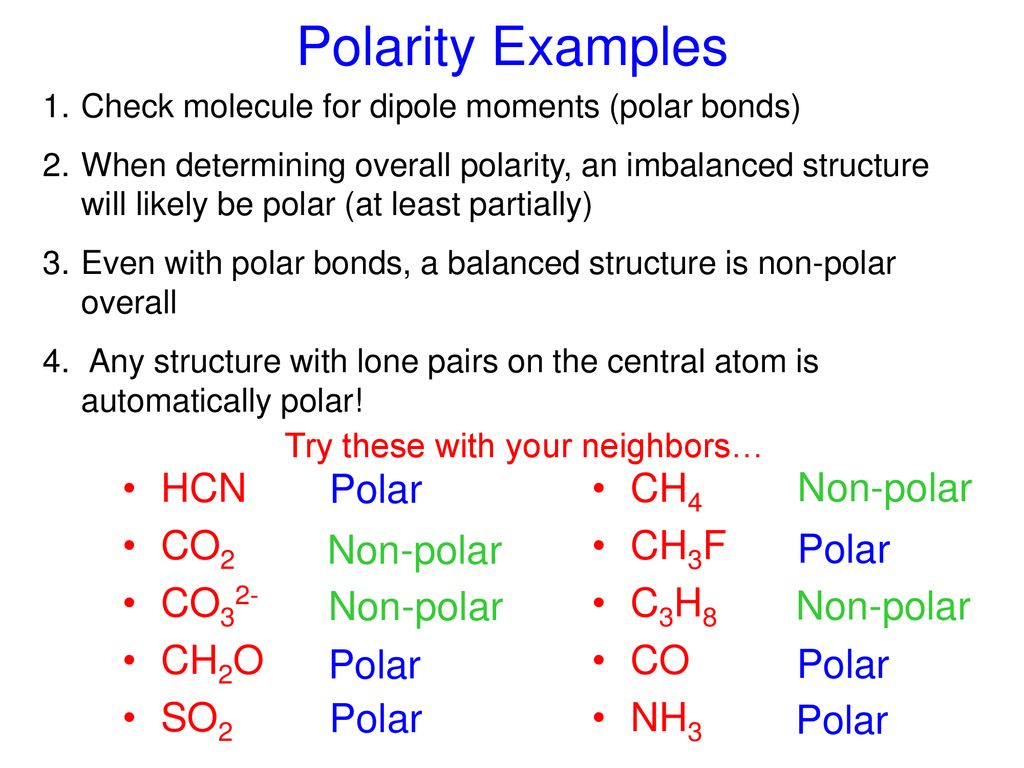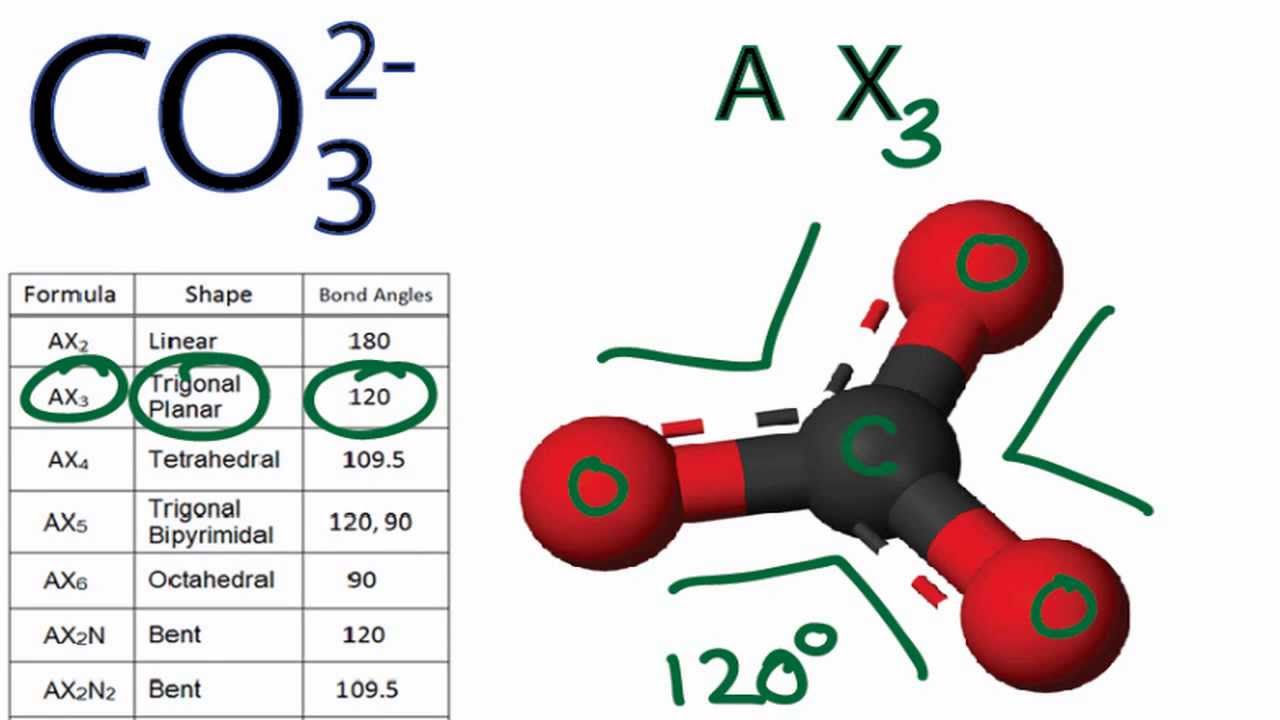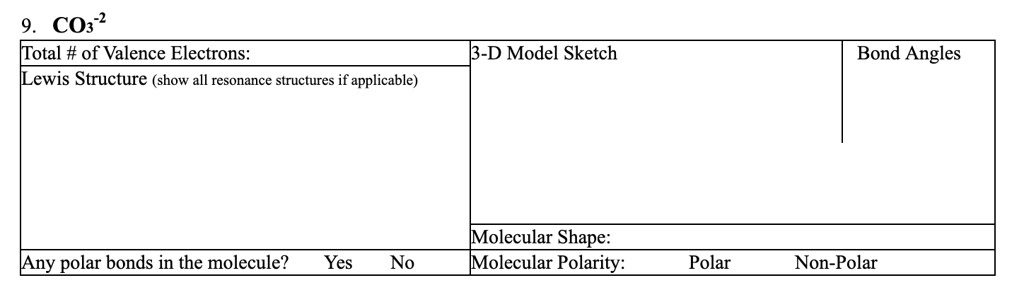
SOLVED: CO32- Total # of Valence Electrons: 24 Lewis Structure (show all resonance structures if applicable) 3-D Model Sketch Bond Angles Molecular Shape: Trigonal Planar Molecular Polarity: Non-Polar Any polar bonds in

NH2- Lewis structure, molecular geometry or shape, electron geometry, bond angle, hybridization | Molecular geometry, Molecular, Covalent bonding

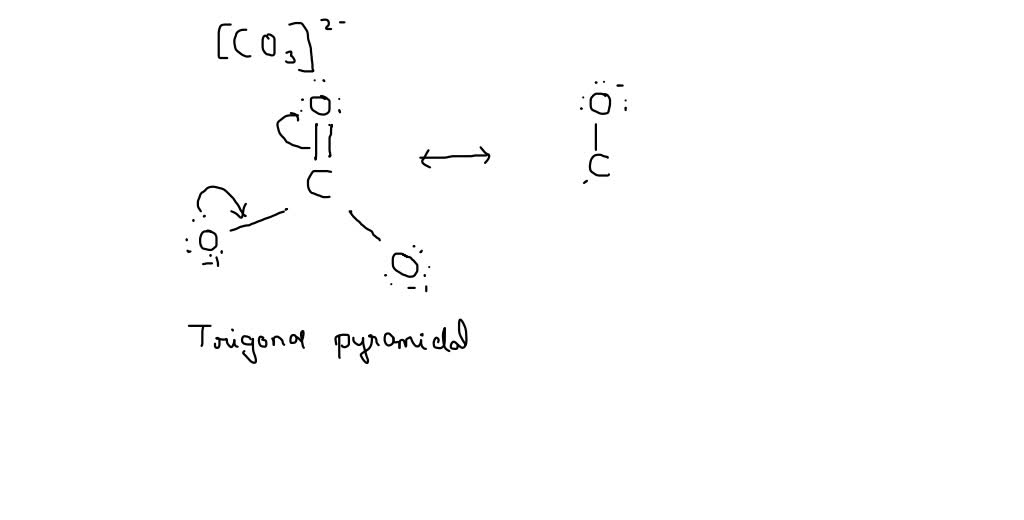
CO32- Lewis structure, molecular geometry, bond angle, formal charge, hybridization | Molecular geometry, Molecular, Electron configuration
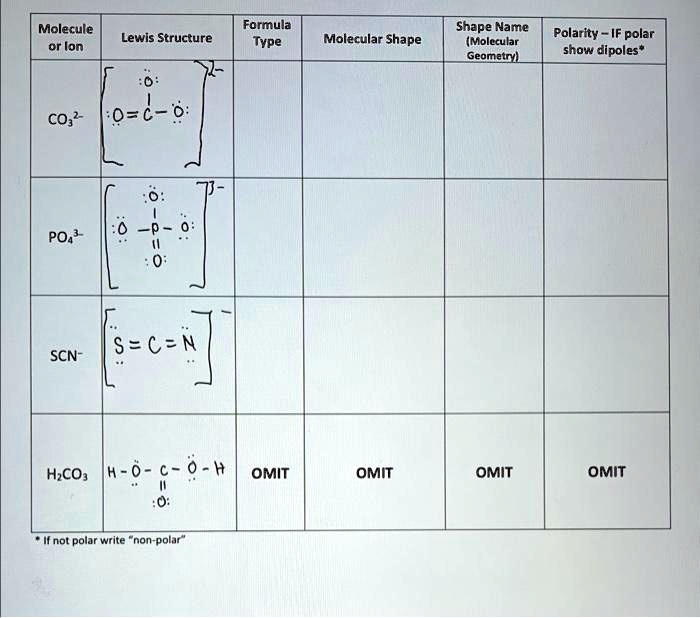
SOLVED: Molecule Formula Type Molecular Shape Shape Name (Molecular Geometry) Lewis Structure Polarity - IF polar show dipoles* 0 CO2 O=C=O 0=d- 0=d- 1 0 PO43- N≡S SCN- HCO2- H-O-H 1 1

Determine if each compound or ion below has a dipole moment. (a) Carbonate ion (CO_3 ^{2-}) | Homework.Study.com