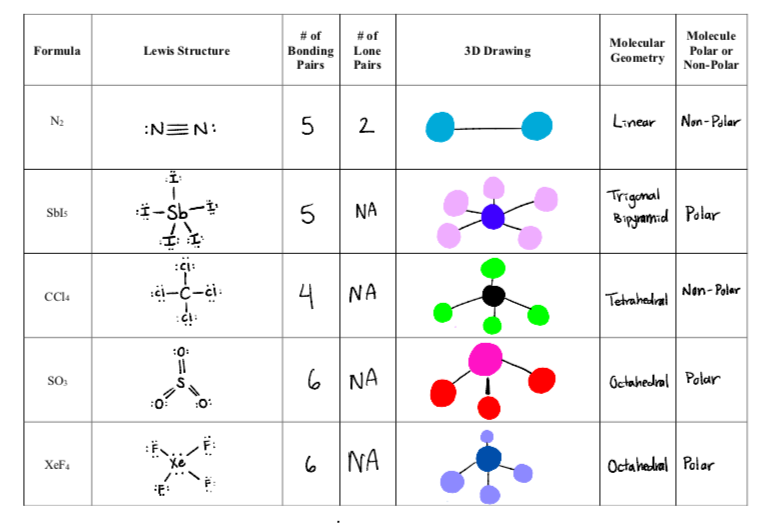
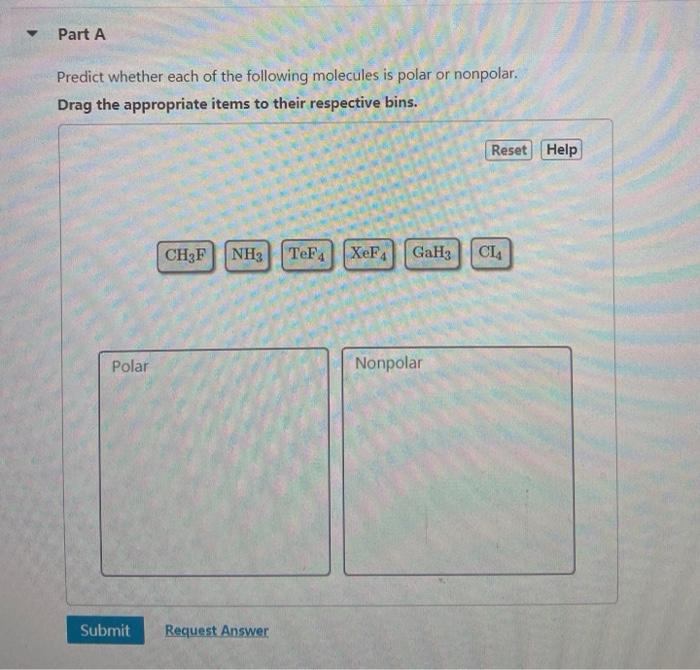
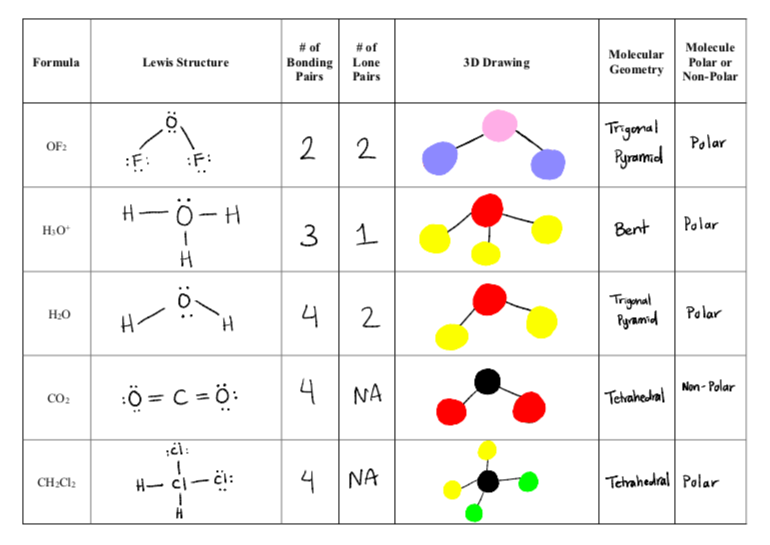
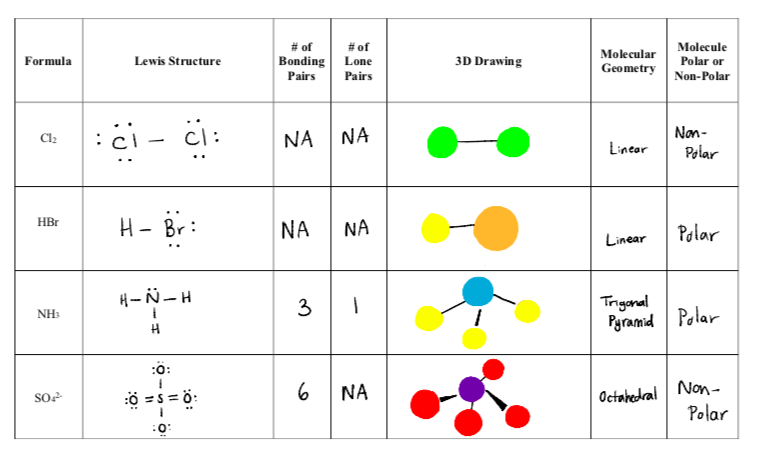
Which of these molecules is the most polar? a. CH3Cl b. C2H6 c. CH3CHO d. CO2 e. all are nonpolar | Homework.Study.com

SOLVED: Choose the compound below that contains at least one polar covalent bond, but is nonpolar: GeH2Cl2, Si2, AsF5, CBr2Cl2. All of these are nonpolar and contain a polar covalent bond.
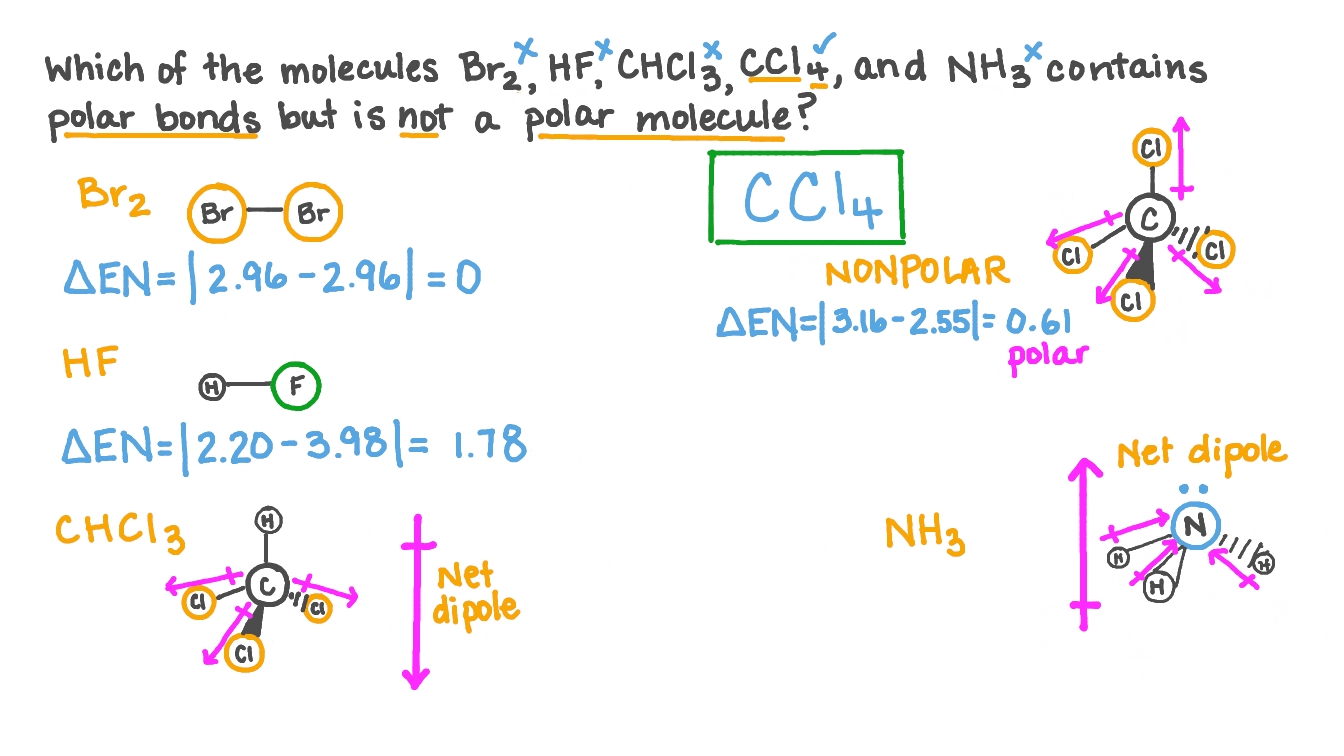
Question Video: Determining the Molecule That Contains Polar Bonds but Is Not a Polar Molecule | Nagwa

Which of the following molecules can be nonpolar? (Select all that apply.) - cis-CHBr=CHBr - CH2=CH2 - cis-CHCl=CHCl - trans-CHCl=CHCl - none of the above | Homework.Study.com

Is SiF4 Polar or Non-polar? (Silicon Tetrafluoride) | Is SiF4 Polar or Non-polar? (Silicon Tetrafluoride) In today's video we are going to help you determine the polarity of the SiF4 molecule. It

Chemical Bonding: Objectives: Illustrate chemical bonding using Lewis Dot structures. Establish chemical formula and name of ionic compounds. - ppt download

Periodic Table Groups 1. Color-code the following groups on your periodic table: -alkali group -alkaline group - transitional group -halogen group -noble. - ppt download
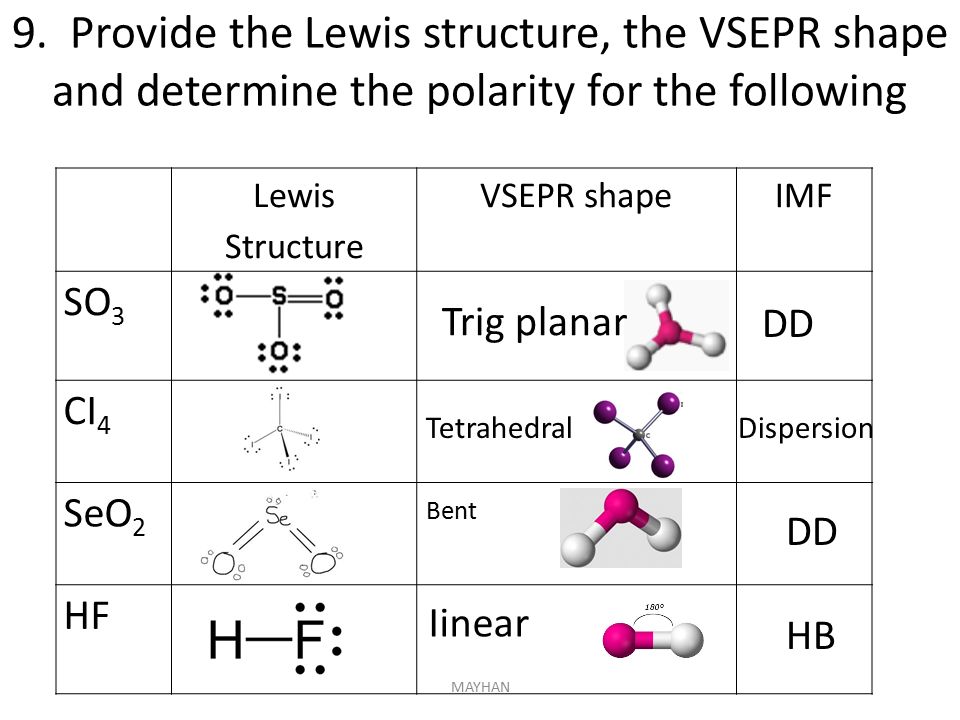
MAYHAN Ch. 7-8 Review Sheet. 1. What are the properties of IONIC substances? These substances: -Solid Hard and brittle (like salt) at room temp -Conduct. - ppt download
Which compound among CCL4 (carbon with chlorine) and CI4 (carbon with iodine) is more covalent and why? - Quora