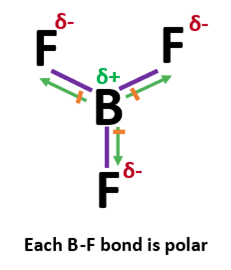
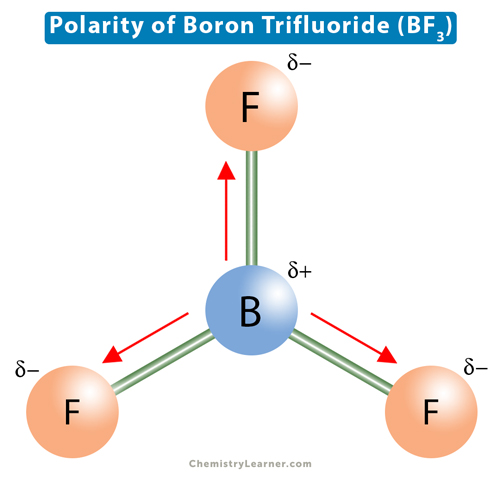
All three of the boron-fluorine single bonds in bf3 are polar. in which direction should the polarity - brainly.com
Both BF3 and NF3 are covalent compounds but NF3 is a polar compound while BF3 is non-polar. How can you explain it? - Quora

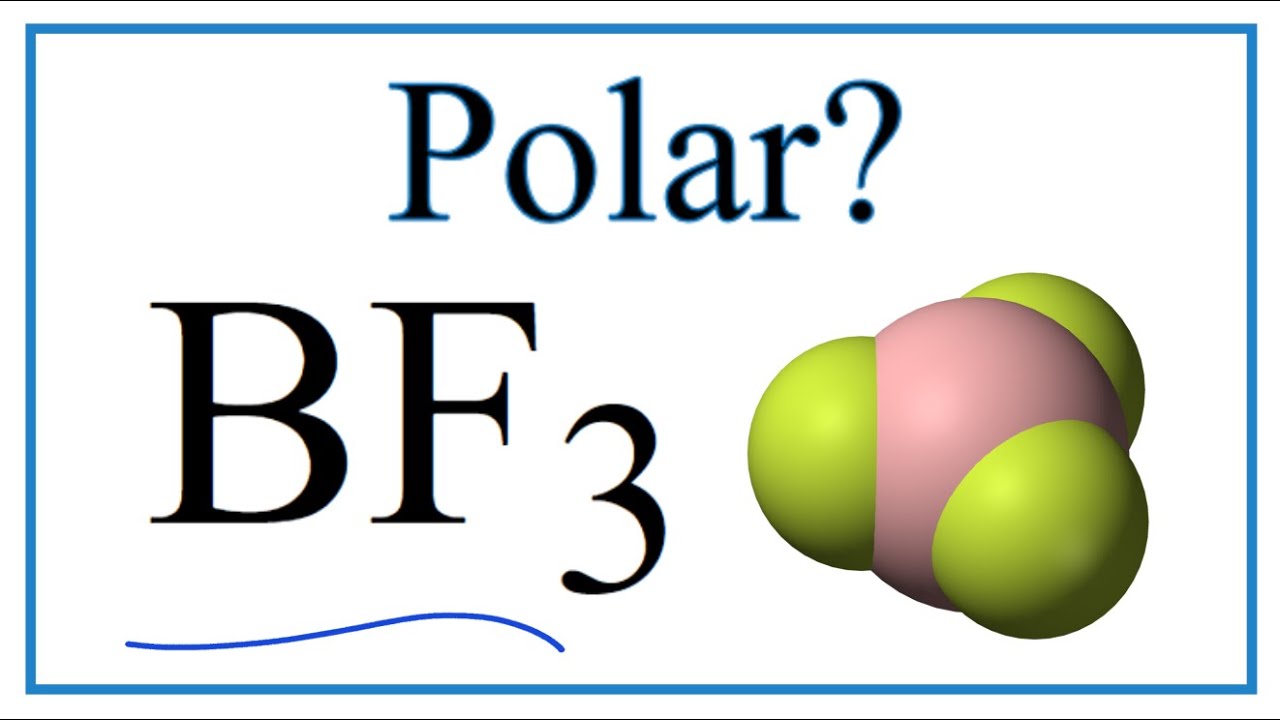
BF3 lewis structure, molecular geometry, polar or nonpolar, hybridization, Bond angle | Molecular geometry, Molecular, Vsepr theory
Boron trifluoride (displaystyle B{ F }_{ 3 }) is a nonpolar molecule, whereas ammonia (displaystyle N{ H }_{ 3 }) is a polar molecule. The difference in polarities is related to the

Si definisce dipolo elettrico un sistema costituito da due cariche elettriche puntiformi q, dello stesso valore ma di segno contrario, vincolate tra loro. - ppt scaricare
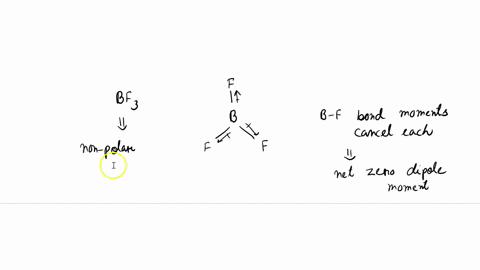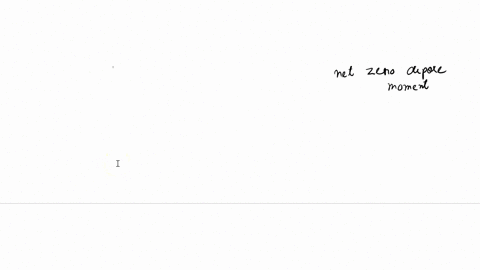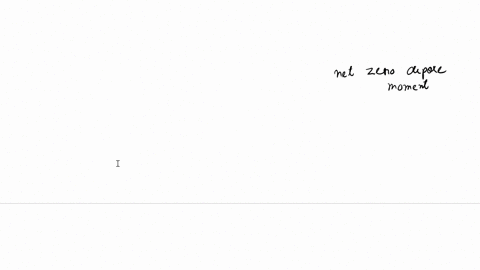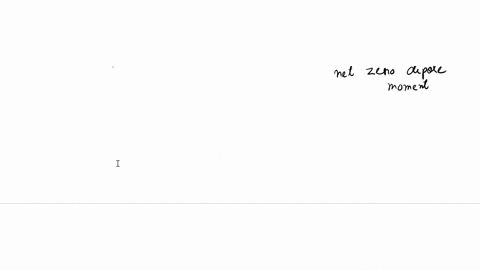


![ANSWERED] All three of the boron-fluorine single bonds in BF3 are - Kunduz ANSWERED] All three of the boron-fluorine single bonds in BF3 are - Kunduz](https://media.kunduz.com/media/sug-question/raw/59983136-1659772204.1988795.jpeg)